
September 2019
The ASPHO Advocacy Brief
Welcome to the Advocacy Brief, a quarterly digital offering from the American Society of Pediatric Hematology/Oncology. The digital publication is delivered directly to Society members' inboxes.
*Send your health policy and advocacy news to This email address is being protected from spambots. You need JavaScript enabled to view it. .
ASPHO Bill Tracker
Visit ASPHO’s Legislative Tracking Chart to see a list of bills that ASPHO is monitoring.
Special Feature Contributions
Sickle Cell Disease: Advocacy Begins in the Clinic
By Lori Luchtman-Jones, MD, ASPHO Advocacy Committee Member
It’s back to school time, and if, like me, you care for inpatients on a pediatric hematology service, it means a spike in sickle cell vaso-occlusive crisis admissions. Did you know that sickle cell disease (SCD) is the fifth most common diagnosis among Medicaid-insured people with four or more hospitalizations per year? That is just one of the thought-provoking statistics in a recent publication by D.C. Brousseau et al. (Pediatrics.2019;144:e20183285). The authors used a multi-state database of Medicaid claims to analyze hydroxyurea prescriptions filled for children (ages 1 to 19 years) with sickle cell disease over the years 2009-2015.
Medicaid insures about 80% of children with sickle cell disease. Among about 5,000 children with sickle cell disease, the percentage with at least one hydroxyurea prescription filled rose from 14.3% in 2009 to 28.2% in 2015 (p<.001). Looking just at HbSS disease, this increased from 18.1% to 37.8%. A little less than half of these children had hydroxyurea prescriptions covering at least 66% of their insured days. The beneficial effects of hydroxyurea in reducing sickle-related crises are widely accepted, but only about one in five of the kids with HbSS had medication available for at least 66% of the year in 2015.
This newsletter is devoted to advocacy efforts, which usually means learning how we can motivate our politicians to improve the health of our youngest and most vulnerable patients. When it comes to sickle cell patients, though, our advocacy efforts need to start in our own clinics to improve access to hydroxyurea and to promote better medication adherence.
Promoting Blood Donation
By Shep Nickel, MD, Vice-Chair of ASPHO Advocacy Committee
As hematology/oncology providers, we know that blood transfusions are life-saving for children with many different diagnoses, but it is important to recognize that patients with sickle cell disease have unique transfusion needs. Given their increased risk for alloimmunization, clinical guidelines recommend that patients with sickle cell disease receive red blood cell transfusions matched for certain minor antigens (C/c, E/e, K). 1) Patients who develop red blood cell antibodies typically need even more extensive matching. This matching has major implications on blood donation requirements. African American donors are more likely to match patients with sickle cell disease. For example, while over 50% of patients with sickle cell disease and African American donors are D+C-E-K-, less than 3% of whites have this phenotype. A recent study projected that 55 African American donors per day would meet 95% of sickle cell disease patient need, whereas 300 white donors per day would meet only 80% of this need. 2) And, as we move towards genotype matching in transfusion, the need for African American donors will further increase.
So how can you help with this issue? While it is very worthwhile to promote blood donation for all, there is a specific need to promote blood donation among African Americans. As a medical professional, you can educate the community about why African American donors are especially needed for patients with sickle cell disease. A few specific things you can do to help promote donation:
- Contact a local radio or TV station to ask if they could do a story on blood donation and sickle cell disease.
- Write a letter to your local newspaper about the importance of blood donation for sickle cell disease.
- Ask to speak at a community event or religious gathering about blood donation and sickle cell disease. An educational program involving African American churches significantly increased the rate of first-time blood donors.
- Host a blood drive. If your hospital does not have a donor center, use AABB’s blood center locator available at http://www.aabb.org/giveblood or contact an organization like the American Red Cross (https://www.redcrossblood.org/) or America’s Blood Centers (https://americasblood.org/).
- Post on social media. Share a fact or statistic about the need for African American blood donors for sickle cell disease.
- Encourage motivated patient families to get involved. Our hospital started a “Blood Donor Ambassador” program for patients and their families to promote blood donation in their community.
- Utilize resources available through community groups, such as the RedHHoTT project led by the Georgia Health Policy Institute (http://mysleevesup.com/about). In addition, contact organizations engaged in efforts to increase blood donor diversity, such as the Sickle Cell Disease Coalition working group on blood donor diversity (http://www.scdcoalition.org/).
References:
- Yawn BP, Buchanan GR, Afenyi-Annan AN, et al. Management of sickle cell disease: summary of the 2014 evidence-based report by expert panel members. JAMA. 2014; 312(10):1033-1048.
- Chou ST, Evans P, Vege S, et al. RH genotype matching for transfusion support in sickle cell disease. Blood. 2018; 132(11):1198-1207.
- Price CL, Johnson MT, Lindday T, et al. The Sickle Cell Sabbath: a community program increases first-time blood donors in the African American faith community. Transfusion. 2009; 49(3):519-523.
Special Update: What Physicians Should Know About Hydroxyurea Use in Pediatric Sickle Cell Disease
By Lori Luchtman-Jones, MD, ASPHO Advocacy Committee Member
At the end of 2017, the Food and Drug Administration approved hydroxyurea for pediatric sickle cell patients over the age of 2. As more clinicians use hydroxyurea for pediatric patients, there are some things to know.
Benefits of hydroxyurea:
- Increased hemoglobin F
- Fewer vaso-occlusive events (pain, acute chest syndrome)
- Reduction in hospitalizations
- Fewer transfusions
- Lower transcranial doppler (TCD) flow by 15-30 cm/second
- Improved survival
- Improved outcomes regardless of clinical severity
Before, during and after with ongoing use of hydroxyurea in a HbSS patient (thanks to Patrick McGann, MD, for the photos)
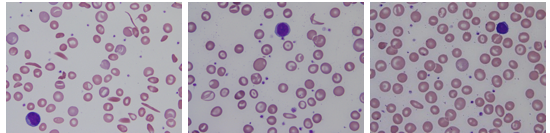
Emerging information in the literature:
- Start at 15-20 mg/kg/day
- Dose escalate every 1-3 months based upon CBC, white blood cell differential and reticulocyte count to maximally tolerated dose
- Precision medicine/pharmacokinetic-based dosing is being studied in clinical trials.
- Infants treated at 6-12 months in clinical trials seem to have a higher and more sustained increase in HbF.
Unanswered questions for hydroxyurea use:
- Long-term safety and efficacy (though thousands of patients have been treated since the 1990’s)
- Use during pregnancy
- Efficacy and safety in other than sickle cell anemia (HbSS, HbSB0 thalassemia)
What's New with ASH Advocacy?
By Connie Piccone, MD, ASPHO Advocacy Committee Member and Liaison to ASH Government Relations Committee
Physicians, particularly pediatricians, can play a powerful role in creating lasting and meaningful change in the lives of the patients whom they serve. Patient advocacy is a priority for our profession, whether at the individual, community, state, or federal level. As more physicians become actively involved in advocacy, policy makers and community leaders will better understand that we have expertise in child health policy issues. The American Society of Hematology (ASH) has a long-standing commitment to advocacy in the advancement of research and treatment for blood disorders, including sickle cell disease. The Sickle Cell Disease Association of America (SCDAA) announced the formation of the SCD Congressional Caucus in 2014. The Caucus is charged with bringing public and congressional awareness to the unique needs of the SCD community in order to address barriers in access to and development of crucial treatments.
There are many ways to deliver messages, ranging from calling your legislator’s office or writing a letter to meeting your Senators or Representative or attending a townhall meeting. Below is a set of practical tools and actions:
- Emailing, calling, or tweeting Congress
- Meeting with your elected officials (face-to-face meetings on Capitol Hill or local Town Hall meetings)
- Advocacy tools (fact sheets, informational handouts or graphics, talking points)
- Newspaper articles (i.e. editorials)
ASH is undertaking a multifaceted initiative ('Sickle Cell Disease Initiative') to address the burden of SCD, both nationally and globally. Throughout the initiative, ASH has engaged a broad array of stakeholders to identify the highest priorities needed to improve outcomes for individuals with SCD. Initiative priorities include:
- Sickle Cell Disease Coalition (SCDC), founded in 2016 'to help amplify the voice of the SCD stakeholder community, promote awareness, and improve outcomes for individuals with SCD'.
- SCD Research Priorities
- ASH Research Collaborative (RC), established in 2018 to foster collaborative partnerships to accelerate progress in hematology. The foundation of the Collaborative is its Data Hub, a technology platform that facilitates the exchange of information by aggregating in one place, and making available for inquiry, research-grade data on hematologic diseases. The first research initiative of ASH RC is a SCD Clinical Trials Network (CTN), which will launch this year to optimize the conduct of clinical trials research in SCD.
- Access to Care
- Policy Priorities
- Global Issues
- Sickle Cell Trait Resources
Additionally, ASH also created the ASH Advocacy Leadership Institute in 2011 to provide additional opportunities for ASH members to learn more about advocacy, health policy, the legislative process and to become engaged in the Society’s activities. This two-day workshop is an opportunity for ASH members to gain a better understanding of the Society and to learn about legislation and health policy affecting hematology research and practice.
If you are interested in learning more about the Sickle Cell Disease Initiative, please contact Sickle Cell Disease Policy and Programs Coordinator Karina Ngaiza at This email address is being protected from spambots. You need JavaScript enabled to view it. .
Visit the ASH Advocacy Center at https://www.hematology.org/advocacy/ for more information on advocacy.
References:
https://www.aap.org/en-us/advocacy-and-policy/Pages/Advocacy-and-Policy.aspx
https://www.sicklecelldisease.org/project/the-president-signs-the-sickle-cell-treatment-act-of-2018/
https://www.hematology.org/Advocacy/4329.aspx#SCDcoalition
https://www.hematology.org/Advocacy/ALI.aspx
Trivia Question #1 (Courtesy of Patrick McGann, MD)
Regardless of ethnicity, what is the most commonly detected disease on newborn screening in the United States?
- Congenital hypothyroidism
- Sickle cell disease
- Phenylketonuria
- Cystic fibrosis
- Maple syrup urine disease
Answer: B – sickle cell disease Sickle cell disease is diagnosed in approximately 1/2000 babies born in the United States (and about 1/350 in Africa). All 50 states now offer universal screening for sickle cell disease. Early diagnosis improves survival through initiation of penicillin prophylaxis, immunization and parental education about the complications of sickle cell disease.
Vichinsky E, Hurst D, Earles A, Kleman K, Lubin B. Newborn screening for sickle cell disease: effect on mortality. Pediatrics. 1988;81:749-55. PMID 3368274
Trivia Question #2
Can you name the first drug approved by the Food and Drug Administration (FDA) for treatment of acute crises in severe sickle cell anemia?
Answer: In 1997 the FDA approved hydroxyurea for use in adults with sickle cell anemia who had at least three pain crises annually. It wasn’t until 2017 that its use in children was approved by the FDA. 2017 was also the year for approval of L-glutamine oral powder to reduce painful crises in adults and children with sickle cell disease.
For more information on our coalition partners and activities here are some helpful links:
American Society of Hematology
American Academy of Pediatrics
Please visit our Health Policy & Advocacy website section for past issues of Advocacy Brief.
