The ASPHO Conference: Advancing knowledge, discovery, and collaboration
The premier pediatric hematology/oncology meeting, the ASPHO Conference brings together 1,300 professionals from around the world each year to gain critical subspecialty education, network with colleagues, and discover innovations advancing the field.
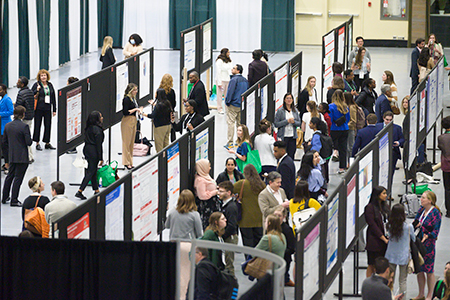
Attendees receive superior educational programming from internationally known experts in the field of pediatric hematology/oncology - 92% of conference evaluation respondents noted they will be able to directly apply conference education to their professional practice and job function. The educational programming includes a dynamic variety of symposia, workshops, plenary sessions, and poster sessions.
Whether you are a practitioner, fellow, faculty member, hospitalist, resident, medical student, physician assistant, or any other advanced practice provider in the pediatric hematology/oncology field, the ASPHO Conference is where you need to be!
“I’ve been coming to ASPHO for 10 years because it allows me to catch up on all the latest and greatest scientific breakthroughs and the more I learn the more I make my patients lives better.” - Conference Attendee
Conference Dates
2026 ASPHO Conference
April 29-May 2
Minneapolis Convention Center
Minneapolis, MN
2026 Call for Workshops
May 7 - July 31, 2025
2026 Call for Abstracts
October 2 - December 18, 2025
2026 Call for Late-Breaking Abstracts
January 5 - January 29, 2026
2027 ASPHO Conference
May 11-14
Palais des congrès de Montréal
Montreal, Québec, CA


